Resources
The Marcus Institute makes available a number of resources for the research community, including data sets and a clinical trial handbook.
At the Hinda and Arthur Marcus Institute for Aging Research, we encourage collaboration and knowledge sharing. Our shared resources are available not only to Marcus Institute faculty but also to our external community of research collaborators and investigators.
The Marcus Institute is uniquely poised to lead the design and conduct of clinical trials in older populations. The expertise at the Marcus Institute is well-suited to trials of cognitive, behavioral, functional, nutritional, technological, and multicomponent interventions in vulnerable older persons in various health care settings. Properly conducted, these trials will directly inform clinical and policy decisions pertaining to the health of older persons.
Our team supports interventional studies that are being proposed and conducted primarily by Marcus Institute faculty. We support traditional clinical trial designs, but have particular expertise conducting trials testing non-pharmacological, multicomponent interventions and that use cluster randomization. We also focus on conducting placed pragmatic trials embedded into the heath care systems or settings that care for older persons
Our aim is to streamline research processes so trials are completed and reported as rigorously and efficiently as possible, and in full compliance with ethical and regulatory standards.
Activities include:
- Consultation for design and conduct of clinical trials
- Collaborative grant review and preparation for proposals involving clinical trials
- Biostatistical design and analytic support for ongoing Marcus Institute projects and applications in development
- Technical assistance including development of protocols and manuals of operation, compliance reports, database collection tools, and data management for active trials of non-pharmacological intervention trials in older persons.
- Provide junior investigators and project staff with rigorous training in interventional research design and data analysis of clinical and quantitative scientists, with emphasis on the epidemiology and treatment of illnesses of aging.
In order to advance the quality of research involving older adults, we’ve taken what we’ve learned from extensive experience in clinical trial design and execution and created training tools that are available to the resesarch community.
Clinical Trials Handbook
Guidance for Clinical Trial Start-Up Period
The NIH Data Management and Sharing (DMS) policy promotes the sharing of scientific data.
Sharing scientific data accelerates biomedical research discovery, in part by enabling validation of research results, providing accessibility to high-value datasets, and promoting data reuse for future research studies.
Marcus Institute Data Archive
Marcus Institute highlighted datasets:
This tool applies the standard deficit-accumulation approach to routinely collected items of a comprehensive geriatric assessment to calculate the frailty index.
Claims-Based Frailty Index
This index estimates a deficit-accumulation frailty index using International Classification of Diseases diagnosis codes, Current Procedural Terminology codes, and Healthcare Common Procedure Coding System codes in the prior 12 months in administrative claims data. It is available for SAS, Stata, R, Python, and Java.
Dr. Pascual-Leone’s Brain Health research team has developed the Home-based transcranial electrical stimulation (HB-tES) Training and Supervision Program for other research groups to teach laypeople how to administer tES in a home setting. The need to properly train individuals in the administration of HB-tES is essential, and there is currently a lack of a uniform training approach across research studies. Here, we present our training program: a tele-supervised, instructional, and evaluation program to teach laypeople how to administer HB-tES to a patient or participant and provide a standardized framework for remote monitoring of participants by research staff.
Many tES research and clinical centers currently involve frequent travel for patients or participants to receive treatment. Home-based tES, however, allows for the therapeutic intervention to occur in the comfort of their own home, administered by a caregiver, family member, friend, etc. This eliminates the need for frequent travel and lessens the mental, physical, and socioeconomic burdens placed on participants and their loved ones. This is especially true for older adults with greater functional limitations, and why we aim to make this intervention as accessible as possible for other researchers and clinicians looking to adapt HB-tES for their own needs.
The use of the HB-tES Training and Supervision Program and its materials is intended for research groups to use in the context of their IRB-approved studies. We do not endorse its use for other purposes or by laypeople using a tES device independently without research staff supervision.
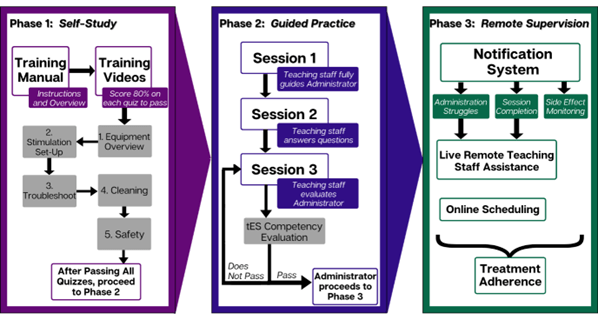
Below is a representation of the three phases in our HB-tES Training and Supervision Program:
- Self-Study: We provide a set of training videos, quizzes, and a manual to help familiarize participants and administrators.
- Guided Practice: Through three practice sessions done remotely, research staff teach laypeople how to administer the HB-tES, and finally evaluate them using a competency checklist.
- Remote Supervision: While the participant and their administrator complete the HB-tES sessions independently, research staff are available remotely to assist with difficulties, monitor side effects, and schedule sessions.
Below, we have all the necessary training materials you will need to implement our training program for your own study or clinic. In addition, our publication which clearly outlines the use of our HSL HB-tES Training and Supervision Program in the context of several pilot studies. Please note that these are examples and should be adapted by other research laboratories to fit within their IRB-approved study parameters and use of different HB-tES devices. We do not endorse any specific HB-tES devices or companies.
Training Manual
Quiz Questions
Video Narrative
Competency Checklist
For any questions, please contact our lab at homestim@hsl.harvard.edu.
You can now learn from the Hinda and Arthur Marcus Institute for Aging Research’s latest findings in a series of guides that address issues faced by older adults and their families.
Available for download:
The Delirium Guide provides information about delirium for patients, family members, and caregivers. Download this brochure to learn more about delirium, including tips for reducing the risk of delirium in the hospital and how to help care for a loved one who is experiencing delirium.
“Preventing Falls in Older Adults” contains evidence-based information for older adults, family members, and health care providers. Our goal is to raise awareness of known risk factors for falls in older adults and practical steps to reduce these risks.
“Advanced Dementia: A Guide for Families” provides information to the family members and individuals responsible for making health care decisions for patients with advanced dementia. The information is intended to complement and encourage, but not replace, counseling by the patients’ direct health care providers.
“How to Talk to Families About Advanced Dementia: A Guide for Health Care Professionals” is a training guide designed for health care providers who support families making treatment decisions for patients with advanced dementia. It’s a companion piece to “Advanced Dementia: A Guide for Families,” which is included.
